 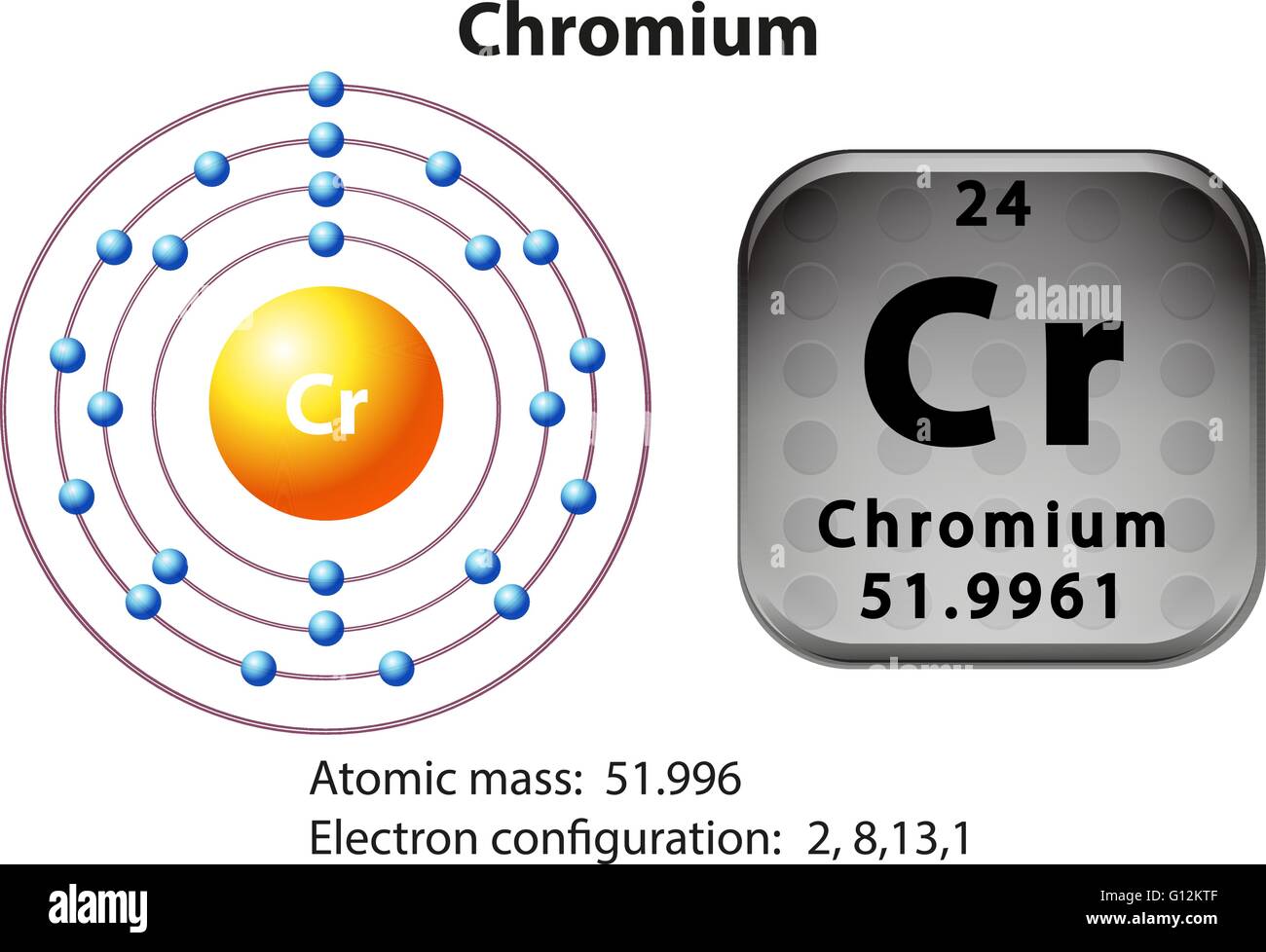
Chromium atom requires one more valence electron in its s-orbital to attain noble. This makes it easier to understand and predict how atoms will interact to form chemical bonds. Coordination complexes of precious metals with the d6 valence electron. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. This give us the (correct) configuration of:įor the Cr 2+ ion we remove one electron from 4s1 and one from the 3d5 leaving us with:įor the Cr 3+ ion we remove a total of three electrons (one from the 4s1 and two from the 3d5) leaving us with Therefore, one of the 4s2 electrons jumps to the 3d5 so that it is half-filled (see video below). Half-filled and fully filled subshell have got extra stability. Therefore we have (still incorrect) 1s 22s 22p 63s 23p 63d 44s 2Ĭorrect Electron Configuration for Chromium (Cr) Potassium, chromium, and copper are the only elements in the fourth period that have one electron in the outermost shell. Both of the configurations have the correct numbers of electrons in each orbital, it is just a matter of how the electronic configuration notation is written ( here is an explanation why). Note that when writing the electron configuration for an atom like Cr, the 3d is usually written before the 4s. 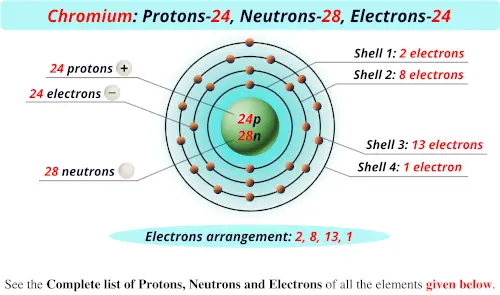
The Co 3+ and Fe 2+ ions, for example, are said to have a d 6 configuration. Therefore the expected electron configuration for Chromium will be 1s 22s 22p 63s 23p 44s 23d 9. Because the valence electrons in transition-metal ions are concentrated in d orbitals, these ions are often described as having d n configurations. After the 4s is full we put the remaining four electrons in the 3d orbital and end with 3d4. In this case, the most energetically stable structure is not the one where the orbitals are half-full. Tungsten has exactly the same number of outer electrons as chromium, but its outer structure is 5d 4 6s 2, NOT 5d 5 6s 1. 
We now shift to the 4s orbital where we place the remaining two electrons. Two rows directly underneath chromium in the Periodic Table is tungsten. Since the 3s if now full we'll move to the 3p where we'll place the next six electrons. We'll put six in the 2p orbital and then put the next two electrons in the 3s. The p orbital can hold up to six electrons. The next six electrons will go in the 2p orbital. Since 1s can only hold two electrons the next 2 electrons for Chromium go in the 2s orbital. In writing the electron configuration for Chromium the first two electrons will go in the 1s orbital. Video: Cr, Cr 2+, and Cr 3+ Electron Configuration Notation 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
